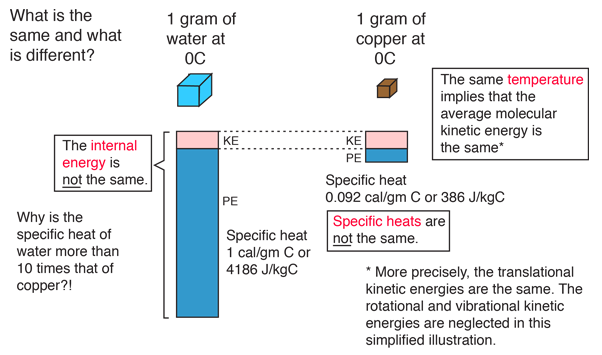
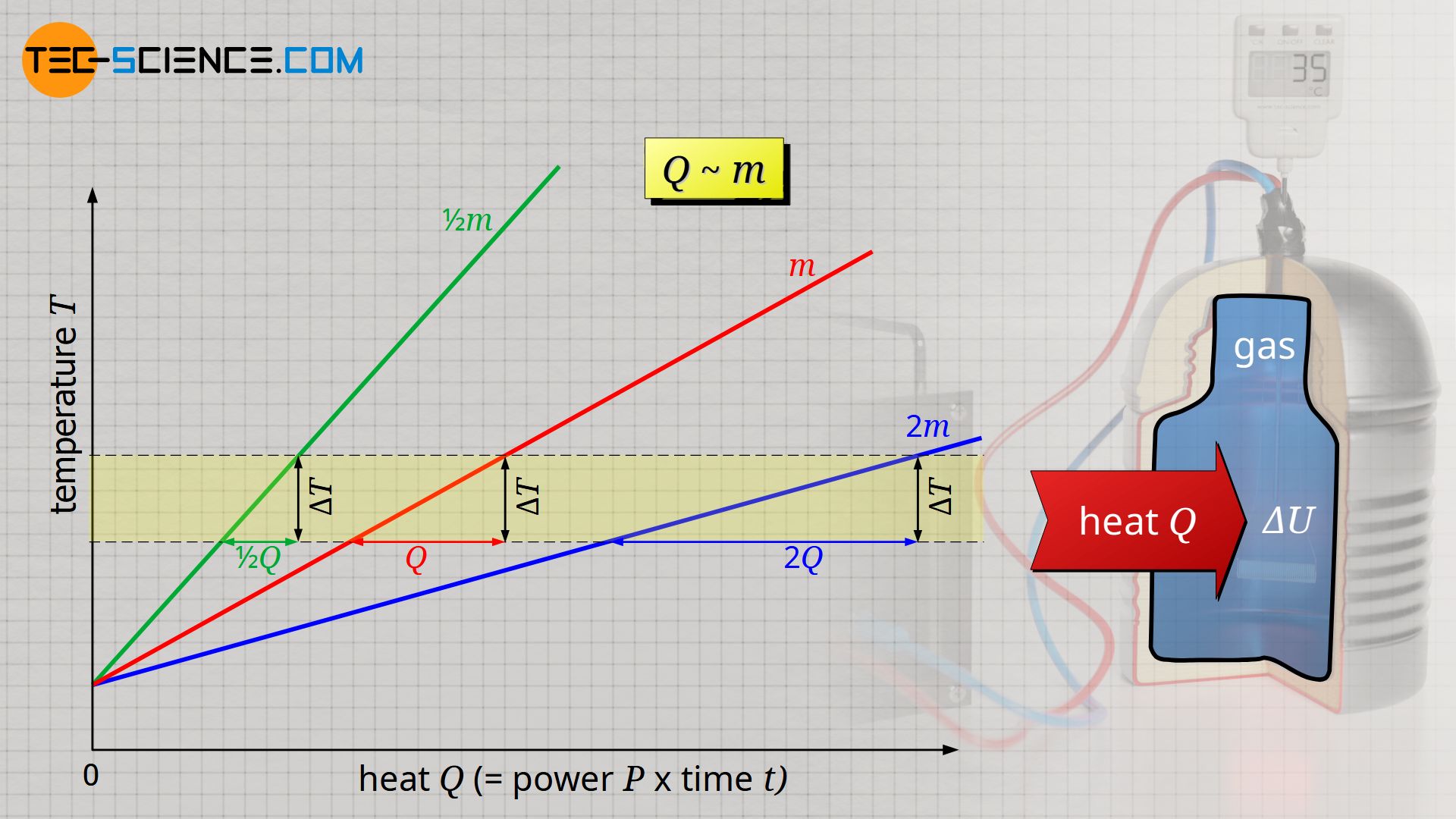
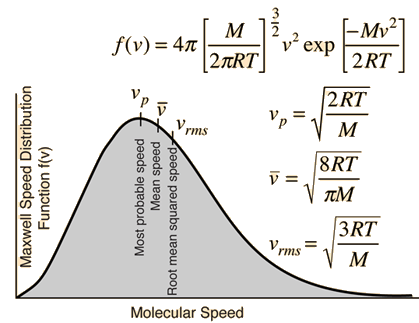
Question Video: Finding the Specific Heat Capacity of a Substance given the Change in Its Temperature and Internal Energy | Nagwa
If temperature is the kinetic energy of atoms, how can metal solids have any temperature if the atoms can't move? - Quora

SOLVED:Calculate the fraction of atom sites that are vacant for copper at its melting temperature of 1084^∘ C(1357 K) . Assume an energy for vacancy formation of 0.90 eV / atom.
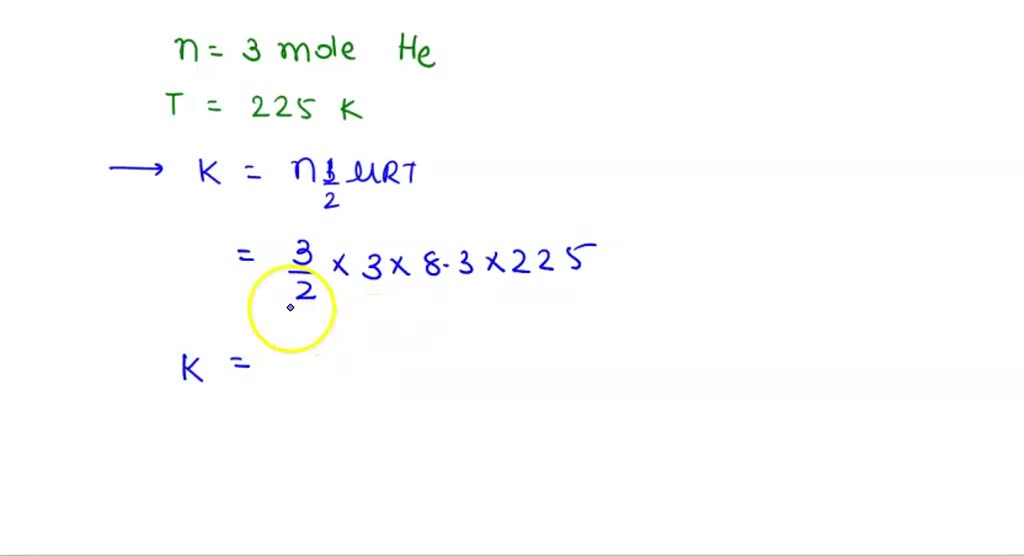
SOLVED: Five moles of a helium gas are at a temperature of 245 K. Calculate the average kinetic energy per atom, the root-mean-square (rms) speed of atoms in the gas, and the

Total energy per atom as a function of temperature. Kinetic energy,... | Download Scientific Diagram

How to Calculate the Average Kinetic Energy of Molecules in Gas at a Certain Temperature | Physics | Study.com

Calculate the increase in energy (in joule) per atom of a piece of aluminium when its temperature is raised by 1^(@)C. Given 27 g of aluminium contains 6xx10^(23) atoms, and specific heat